Diffusion
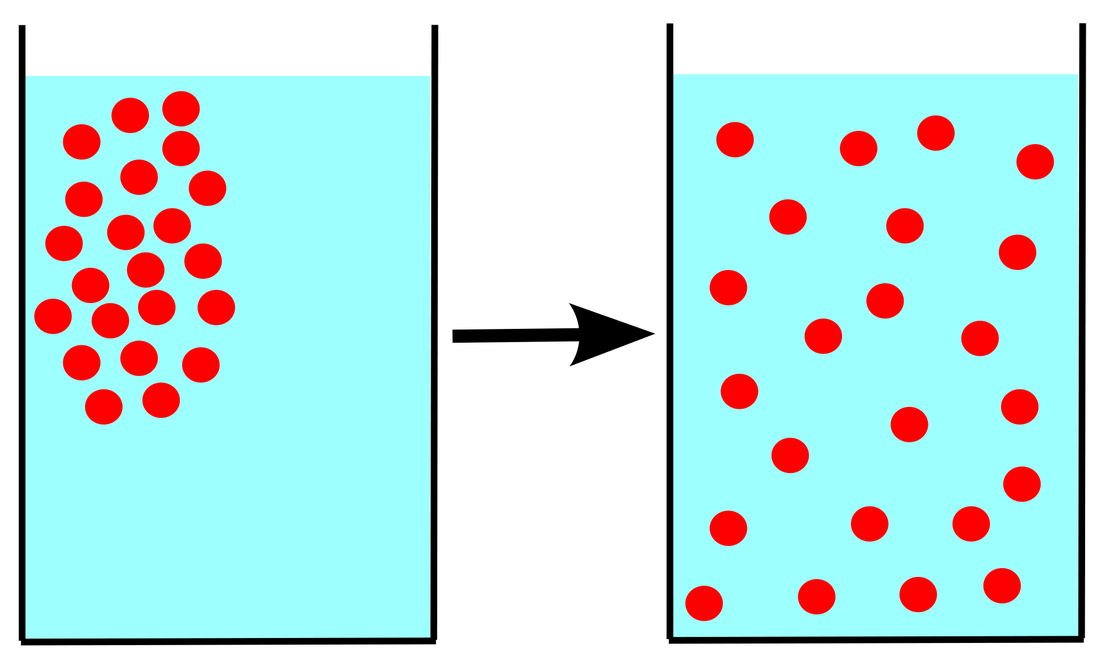
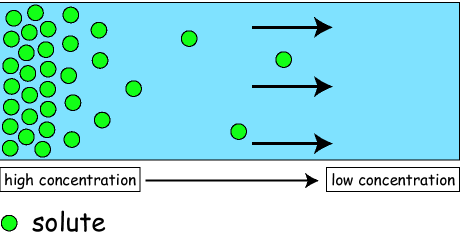
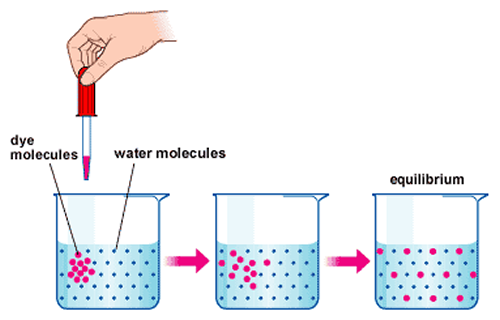
Diffusion is the movement of substance from a high concentration to a low concentration until the substance is in an equilibrium.
Keep in mind that though diffusion would eventually slow down, it will never stop as Brownian motion states that the molecules will move in random directions hence it will never reach an equilibrium.
Diffusion only occurs within a fluid (liquid and gas). One example of this is a salt cube being dissolved within the a glass of water. The salt cube has a high concentration of salt and the water has low concentration. The salt cube will dissolve and the fluid will eventually have an even concentration of salt particles.
Keep in mind that though diffusion would eventually slow down, it will never stop as Brownian motion states that the molecules will move in random directions hence it will never reach an equilibrium.
Diffusion only occurs within a fluid (liquid and gas). One example of this is a salt cube being dissolved within the a glass of water. The salt cube has a high concentration of salt and the water has low concentration. The salt cube will dissolve and the fluid will eventually have an even concentration of salt particles.